
His most inventive solution was that tellurium has a larger than expected atomic mass because it was not prepared in an entirely pure form but contained some of its heavier analog dvi-tellurium, which was the name given to the unknown element that should appear after bismuth in the periodic table. However, this did not solve the problem for a long time the issue of the anomalous atomic mass of tellurium kept returning in his later years as well.  
Mendeleev did not accept the experimentally determined atomic mass for tellurium, but assumed a relatively small error and included 125, as shown in Table 1. Incidentally, tellurium was already displayed before iodine by Julius Lothar Meyer (1830–1895) in his system of elements in 1864. There was only one point where this logic seemed to fail: iodine, which is certainly the chemical analog of bromine and not selenium, had a lower atomic mass than tellurium, whose resemblance to selenium was beyond any doubt. With this arrangement, the chemical (and also some of the physical) properties return periodically so that elements with similar characteristics occupy a single column. The basic idea of the system is that the elements should be arranged in the order of their atomic masses, which were determined by diligent but probably also quite boring scientific work in the preceding half century. Yet the author of this article would like to focus on Mendeleev’s less successful lines of arguments in the hope that they will help the reader to understand the logic behind the work of the great Russian scientist, and also to give some insights into some general questions of scientific thinking. Needless to say, being able to predict unknown phenomena or the existence of unknown substances is one of the strongest arguments in favor of the validity of a scientific theory. This bit of science history is often recalled in textbooks even Wikipedia has a page on the significance of these success stories. 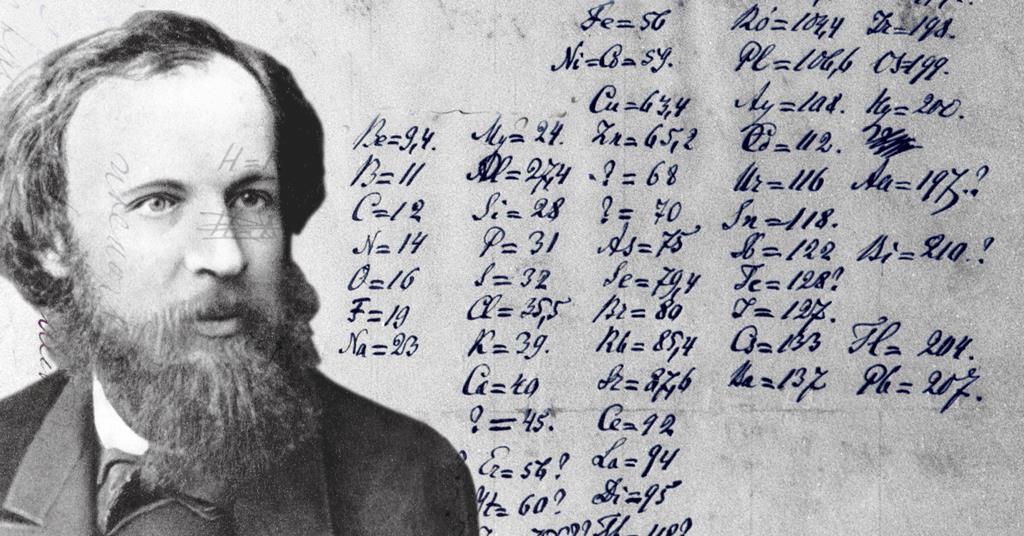
Some of the early predictions were verified within 20 years of the first publication. One of the major reasons why Mendeleev is given most of the credit for developing the periodic table is that he made regular attempts to extract the scientific logic of the system and make verifiable (or falsifiable) predictions based on it. Taking into account the scientific information known in the middle of the 19th century, it is probably fair to say that chemistry was ready for the discovery of the periodic table: there were numerous independent attempts at organizing the elements known at that time, atomic masses were mostly (correctly) determined, and the introduction of the spectroscopic method in 1860 reduced the incidence of false element identifications (although did not eliminate them entirely), which was a major problem hindering any element-systematization work in the first half of the 19th century. Needless to say, the historical aspects are centered mostly on Dmitri Ivanovich Mendeleev (1834–1907), who is remembered both as the uncontested champion of discovering the natural system of chemical elements and the tireless communicator who made the ideas known to the widest possible audience. 2019 is the international year of the periodic table of chemical elements, which provides a good occasion to popularize chemistry through recalling both the scientific principles and the human stories behind the periodic table.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
